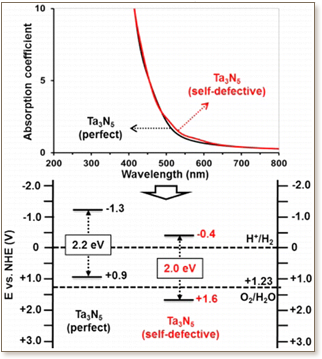
 Relevant properties to visible-light overall water splitting reactions of perfect and self-defective bulk Ta3N5 semiconductor photocatalysts are investigated using accurate first-principles quantum calculations on the basis of density functional theory (DFT, including the perturbation theory DFPT) within the screened coulomb hybrid (HSE06) exchange-correlation formalism. Among the various explored self-defective structures, a strong stabilization is obtained for the configuration displaying a direct interaction between the created N- and Ta-vacancies. In the lowest-energy structure, each of the three created Ta-vacancies and the five created N-vacancies is found to be in aggregated disposition, leading to the formation of cages into the lattice. Although the calculated structural, electronic, and optical properties of the two materials are found to be very similar and in good agreement with available experimental works, their photocatalytic features for visible-light overall water splitting reactions show completely different behaviors. On the basis of calculated band edge positions relative to water redox potentials, the perfect Ta3N5 (calculated band gap of 2.2 eV) is predicted by HSE06 to be a good candidate only for H+ reduction while the self-defective Ta3N5 (calculated band gap of 2.0 eV) reveals suitable band positions for both water oxidation and H+ reduction similar to the experimental data reported on Ta3N5 powders. Its ability to reduce H+ is predicted to be lower than the perfect one. However, the strongly localized electronic characters of the valence band (VB) and conduction band (CB) edge states of the self-defective material only on the N 2p and Ta 5d orbitals surrounding the aggregated N- and Ta-vacancies are expected to strongly limit the probability of photogenerated carrier mobility through its crystal structure.
Relevant properties to visible-light overall water splitting reactions of perfect and self-defective bulk Ta3N5 semiconductor photocatalysts are investigated using accurate first-principles quantum calculations on the basis of density functional theory (DFT, including the perturbation theory DFPT) within the screened coulomb hybrid (HSE06) exchange-correlation formalism. Among the various explored self-defective structures, a strong stabilization is obtained for the configuration displaying a direct interaction between the created N- and Ta-vacancies. In the lowest-energy structure, each of the three created Ta-vacancies and the five created N-vacancies is found to be in aggregated disposition, leading to the formation of cages into the lattice. Although the calculated structural, electronic, and optical properties of the two materials are found to be very similar and in good agreement with available experimental works, their photocatalytic features for visible-light overall water splitting reactions show completely different behaviors. On the basis of calculated band edge positions relative to water redox potentials, the perfect Ta3N5 (calculated band gap of 2.2 eV) is predicted by HSE06 to be a good candidate only for H+ reduction while the self-defective Ta3N5 (calculated band gap of 2.0 eV) reveals suitable band positions for both water oxidation and H+ reduction similar to the experimental data reported on Ta3N5 powders. Its ability to reduce H+ is predicted to be lower than the perfect one. However, the strongly localized electronic characters of the valence band (VB) and conduction band (CB) edge states of the self-defective material only on the N 2p and Ta 5d orbitals surrounding the aggregated N- and Ta-vacancies are expected to strongly limit the probability of photogenerated carrier mobility through its crystal structure.
10.1021/jp506066p